AiA Ep 121 FINAL
===
Intro: [00:00:00] Welcome to AiArthritis Voices 360, the podcast solving today's most pressing issues in the AiArthritis community. We invite you all to the table, where together we face the daily challenges of autoimmune and autoinflammatory arthritis. Join our fellow patient co-hosts as they lead discussions in the patient community, as well as consult with stakeholders worldwide.
To solve the problems that matter most, whether you are a loved one, a professional working in the field, or a person diagnosed with an AiArthritis disease, this podcast is for you. So pull up a chair and take a seat at the table.
Tiffany: Hello everyone and welcome to AiArthritis Voices 360. This is the official talk show for AiArthritis or short for the International Foundation Autoimmune Autoinflammatory Arthritis. And this is [00:01:00] also a combined episode with our special series Rheumy Rounds. Which is when rheumatologists and health professionals come to the table as equals with patients to talk about topics that would improve their healthcare when presented in an office setting.
So we'll touch on that too. So that's why it's a combined, and I am so excited. I am here today with Dr. Vibeke Strand. Hi Vibeke.
Dr. Vibeke Strand: Hi there.
Tiffany: Tell us a little bit about you.
Dr. Vibeke Strand: I teach at Stanford and I've been there for a long time. I also do consulting and new product development and rheumatology, and I've been doing that for a long time too.
And I see patients because I serve as a attending in clinic and I also help teach the fellows, our fellows rheumatology and you know, how they should basically treat their patients and do a good job. It's been very fun and I continue to enjoy doing it, [00:02:00] so that's where I am.
Tiffany: Up. Well, I have no, we've known each other for quite a few years now, is BI is very involved in patient research, partners patient engagement and research.
And so that's where our paths originally crossed. And I'm just so excited that we still connect and are able to have you on the show to talk about important topics like today. Which is vagus nerve stimulation, something that's a super, super hot topic in the patient community. And I got wind that Vibeke's presenting about.
This has been really involved in the groups that are doing research and, and coming out with these vagus nerve modulators. So who better to talk about it? So wanted to invite Vibeke here so we can. Talk more, learn more about what's happening and really just get a lot of important [00:03:00] information so that patients understand what it is.
A little bit about the research and how we got here, and then we'll end with some, how do we talk to our doctors about this? So how does that sound? Ready to go?
Dr. Vibeke Strand: Ready to go?
Tiffany: All right. Take it away.
Dr. Vibeke Strand: Okay, so I'm going to talk to you about vagus nerve stimulated neuro immune modulation for the treatment of rheumatoid arthritis.
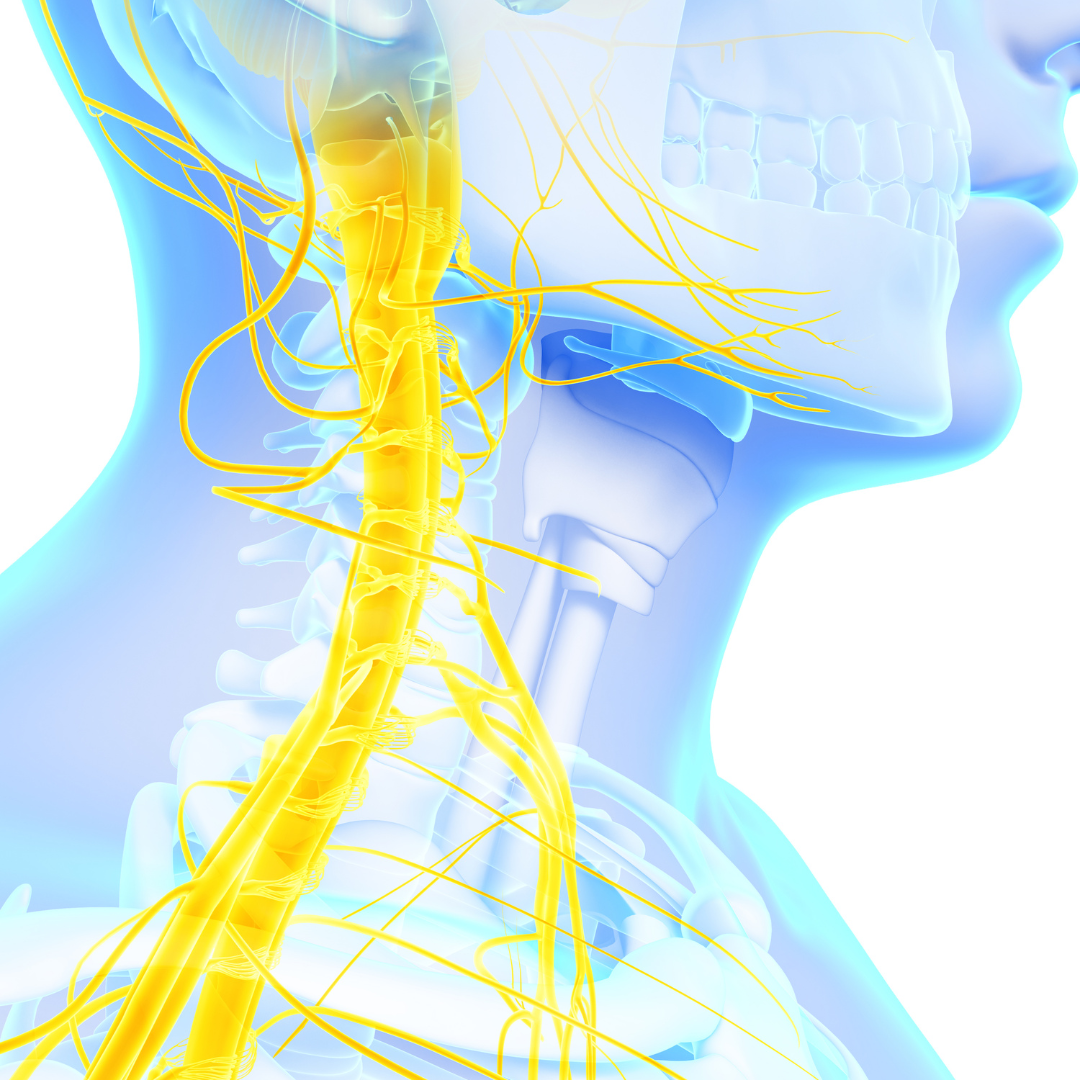
So we start with what we call the neuro immune connection. It's a connection between the vagus nerve and the immune system. So you see the picture and essentially the central nervous system, the brain, the spinal cord regulates the immune system, including through the vagus nerve. The vagus nerve connects the brain to the internal organs in the body, all of the internal organs, and it helps to regulate inflammation.
So basically it both monitors and sends signals to tell the immune [00:04:00] system when to calm down. So research has shown that when the vagus nerve becomes less active. Or after you have what we call a vagotomy, a removal of the vagus nerve, and that used to be done for bad acid reflux disease and stomach problems.
If you have that removed, it could be a risk factor for developing ra, which is interesting and suggests that yes, there really is a connection between the vagus nerve and the immune system. Interestingly enough, vagal nerve stimulation has been used for more than 25 years and has helped more than 150,000 people with diseases such as treatment resistant epilepsy and depression.
Tiffany: So I just wanted to, just really quick, you can, you can keep the slide up. Just, just to reiterate, I don't think as a patient I have heard much. About the vagus nerve as far as how interconnected it [00:05:00] is to, as you said, all of the organs in the body. I mean, that's just absolutely fascinating and I think that the point that you brought up, this isn't new.
And so I think that's important for patients to understand that the ver vagus nerves stimulation has been in research and helping many, many patients. So I just wanted to reiterate. Just the fact that it's such an important piece of our immune system and our body and as a patient, I really didn't understand that.
Dr. Vibeke Strand: Yeah, and I think we haven't recognized that nearly as much. And we've always thought about maybe, if anything the vagus nerve is, is sending down signals or receiving signals, but not that it had much to do with the immune system. And that's what's really interesting.
Tiffany: Absolutely.
Dr. Vibeke Strand: So, as I said to you before, the vagus nerve connects to the major organs and it helps to detect infection or tissue injury.
So that's what we've always [00:06:00] known, and it sends that information to the brain, but it also sends signals from the brain down to help adjust and balance the immune response. And this is called the inflammatory reflex. Since it calms the immune system, naturally we call it, and think of it as an inflammation calming reflex, and you see the picture here and we've got essentially the immune cells where you've got the B cells and then you can see the blood vessels and you've got, basically what's going on is the brain is going down.
It's getting signals up the vagus nerve, the AFA vagus nerve that have the pro-inflammatory signals, the cytokines and what we call the pens. And then going back down, it's sending signals to what we call the celiac ganglion, which goes to the spleen and of course to the lymph nodes, but also to [00:07:00] the intestine and the other internal organs.
And so that's how this system works. So basically when we talk about trying to do vagal nerve stimulation, the application for rheumatologic disease is this electrical stimulation of the vagus nerve. So it triggers that inflammation, calming reflex, which leads to less cytokine production, and the immune cells then produce fewer inflammatory chemicals or cytokines such as TNF, alpha IL six, and IL one.
Which helps to reduce inflammation and essentially a 62nd stimulation can reduce RA inflammation by as much as 30 or 70%. So what you're seeing in the picture is essentially acetylcholine and then the alpha and nitro acetylcholine receptor, and those are the major mechanisms here for down modulating [00:08:00] the T and FIL one IL six, and IL 23.
Tiffany: So let me ask a question here. So when we're talking about the TNFs, the IL six patients do understand someti to, to some extent, that those associated with our biologic drugs. So is this in effect. Help a similar concept to how a biologic may work or that targeting the same things when we're talking about IL six or IL one or IL 23 or TNF?
Dr. Vibeke Strand: Well, it's similar in that it's targeting these cytokines, but it's targeting them together and it's not singularly targeting one and it's not fully inhibiting any of them. It's down, modulating them.
Tiffany: Okay,
Dr. Vibeke Strand: so it's acting more as a modulator rather than inhibitor.
Tiffany: Whoa. Well now that is a very interesting fact.
Thank you. [00:09:00]
Dr. Vibeke Strand: So in addition, with this novel mechanism of action, it can also modulate other anti-inflammatory pathways. And one of the things we know about rheumatoid arthritis and other inflammatory arthritis is it is causes bone damage as well. And so part of the inflammatory process is actually where you get bone resorption.
You lose as much mineralization as you'd like to have, and so you get damage to the bones. And that's how we, we see a lot of the deformity that can occur with rheumatoid arthritis among other things. And so what we found out actually is that vagal nerve stimulation. Because it is again, down modulating these inflammatory processes can in fact support the bone building cells, and those are the osteoblasts and down [00:10:00] modulate the osteoclast, which caused the bone resorption.
What's really nice about that is we've talked about how biologic agents also are helpful for bone effects, and what we see is I'll show, as I tell you, vagal nerve stimulation has a similar beneficial effect on the bone. So now there've been various approaches to stimulate the vagus nerve and. There's direct stimulation, which what we're talking about, which is a small implanted device that's near in the neck, near the vagus nerve, but you can also do other stimulation transcutaneous, and you can do it say from the ear, and that's been done.
But what's been done with this setpoint system that we're talking about? Is first a small study in Europe. It was published in PNAS and then a pilot safety study in the States. It was published in [00:11:00] Lancet Rheumatology, and then this study I'm gonna show you, reset ra, which was a randomized controlled trial in 242 patients at 41 sites in the US with a three month primary endpoint and an extension study to a year, and then they're doing long-term follow up.
This led to approval of this device in 2025, summer of 2025. And this has also been published in Nature Medicine.
Tiffany: So now Viba, let me, let me ask, 'cause I'm looking at the photos here. Of the direct versus a photo that looks like a chip clip on somebody's throat. So that is, so these are kind of, when we're talking about these different approaches, how did the, how did the clip on the throat work out?
Dr. Vibeke Strand: Let me show you.
Tiffany: Okay.
Dr. Vibeke Strand: Here, here is the clip. It's essentially [00:12:00] about the size of a vitamin pill. Okay. And that's it. It is actually implanted in the neck. That takes about an hour to an hour and a half by a neurosurgeon. It's an outpatient surgery, but you have to have general anesthesia. And then after that heals, which is about one to two weeks, and it doesn't heal with any scar, so you don't know it's there.
Then it's activated by your rheumatologist with a programmer app that's on your iPad, and then you'll use the iPad to charge it once a week wearing this implant around your neck and this is how you charge the implant and it's so, it's quite a quite convenient, it's just a few minutes every week. This device lasts for about 10 years.
You can turn it on or deactivated, and when it's deactivated, it's gonna be there, but [00:13:00] it won't be interfering with anything. You can still get an MRI or anything else. So it's not like you have a big device. You don't have any leads from the device anywhere. There's no pockets, there's nothing external. So no one really knows you have it there except you.
And you don't feel it when it's charging, and you certainly don't feel it when it's having its stimulation, which occurs at night, usually for about a minute or two maximum. And you know, the stimulation is adjusted with you to the point where you don't feel it. And it happens at at random times at night.
So it's not something that would ever wake you up. And again, you won't even know it's there.
Tiffany: Wow. So in the evolution of trying to figure out the best ways to modulate the vagus nerve, [00:14:00] the, the kind of, the endpoint where we ended up at was this tiny advice, and you're right, that looks very tiny. And is that about an inch?
Dr. Vibeke Strand: It's not even an inch.
Tiffany: Not even an inch.
Dr. Vibeke Strand: It's literally about the lar size of a vitamin capsule.
Tiffany: Wow. Okay. And so we went from trying kind of the external, you said kind of the clipping of the throat or, or in that kind of thing. Not real. Didn't really work well with the patients. Well, how high dropout rate, how has this been as far as people liking it saying.
Hey, this one, this one works for me.
Dr. Vibeke Strand: It's been very well accepted. In fact, 98% of the people that are in the trial, I'm about to tell you about, finished the trial Wow. For a whole year. Okay.
Tiffany: Okay. Well, tell us more.
Dr. Vibeke Strand: I will. I will. I'd be glad to. So this is a picture of it, and you can see [00:15:00] where it goes in the neck right here.
And you can see relatively, it's quite small. So I said it's good for about 10 years. It can be removed if you want to have it removed. As I said, it's a 60 to 90 minute implantation procedure, which is very simple, and it's done by a neurosurgeon, as I mentioned, and then it's 60 seconds at night sometime.
I just said you can have MRIs without any trouble and you recharge it a few minutes once a week. Okay. So this is just what we learned from this trial. Basically, by 12 months, over 60% of the patients had improvement of a CR 50. Though we have a CR 20% improvements and we have 50% improvements, and that is an improvement that we think is very clinically meaningful, even more so than the 20%.
The MRI data did [00:16:00] show that they were in about. 50% reduction in new joint erosions. In other words, there were no more new damages to the bone. As we had talked about in this predefined high risk subgroup, those that had the most damage at baseline, those are the ones that are most likely to show further damage to their bone.
So it wasn't just symptom relief, it was also really impacting structural damage, which is why we call our DMARDs disease modifying because they offer structural damage prevention or improvement. So by the end of this trial, about 75% of these patients were still using vagal nerve stimulation as their only therapy without additional biologic or targeted synthetics.
Although they were allowed to add them after the three month endpoint and 25% of them [00:17:00] did adding it sometime between three and 12 months, and I said to you, 98% stayed on stimulation has to mean that they found it acceptable and helpful. 'cause I haven't seen a trial where there was 98% completion.
Tiffany: That
Dr. Vibeke Strand: is in a long, long time.
Tiffany: Big number. So, but, so let, let's go over a few of these points here too. So when we talk about the ACR 20 and the ACR 50 being better, that is a measurement essentially that's saying. What we would call like a baseline or where you are right now, you have a 20% improvement or a 50% improvement. Is that a fair way to explain that?
Dr. Vibeke Strand: That's right. And it's a 20 or 50% improvement in both your tender and swollen joint counts, but also in your patient global assessment of disease activity, in your pain and in your hack disability index score or your physical [00:18:00] function. So. What we're looking for is, is five of these seven parameters and the others, the other ones include physician global and either SED rate or CRP five, patient facing measures.
Two physician responses.
Tiffany: Wonderful. Well, that, I think that's really important for patients to really understand too. And they may, an A CR would be the American College of Rheumatology, and so I do think it's important you'll see that if you ever, we have a program called Go With Us to Conferences that some of you out there may ha may go with us.
So when patients like myself go to these conferences. We explain what we're learning and why things like this measure is really important. That's 50% improvement of how you felt when you started. That's a big deal. And so I do think that that's really important to note. And then also I think it's, it's really the, the point that I think what [00:19:00] made me go, what is that about 75 patients were using this as their soul therapy.
No biologic, no disease modifying or dmard because as a patient we have been sort of told, you know, biologics, these are, these pharmaceutical are the, these are really the way that you're going to modulate your inflammation. Make sure you don't get bone erosions, for example. So can you just explain a little bit more?
Is is it's actually showing that some patients are successful? In a, to your point, a different mechanism of action.
Dr. Vibeke Strand: Yes. And I will show you that the safety profile is quite favorable, particularly compared to the biologics and drugs that we've just been talking about, the DMARDs. And there are very few side effects, and there was long-term [00:20:00] safety.
So we've talked about before, increased infections. Malignancies. And then we've also been worried about cardiac safety, cardiac events, particularly with the targeted synthetic DMARDs. And these aren't an issue with this product, but I'm going to show you the data. So this was just the introduction to what the data show you.
Tiffany: Okay.
Dr. Vibeke Strand: So this was a trial. It was 12 months in total. Okay. And the first three months was here, there were 242 patients and they had their implant and then they were randomized either to treatment, which was active stimulation or to sham, which was non-active stimulation. So the device was in, was present, but it was not activated.
Then at three months, everyone was crossed over to get active stimulation [00:21:00] and that was an open label for the rest of the time to the end of the study, which is there, got down here five years, but at month 12 was when the data stopped. There was MRIs done at baseline at month three and and month six that showed.
What we told you about, which was less bone damage. So at three months, all the patients were now having active stimulation, and after three months, patients could add a biologic or a targeted synthetic DMARD if needed. But that was only in the open label period. Okay, so this is the ACR 20 response, and this is up to the three months.
So this is active stimulation to three months, and this is sham treatment, and that is highly statistically significant. The difference was an ACR 20 of [00:22:00] 35% versus 24.2%. Okay, and in patients who had failed more than one biologic or targeted synthetic, in other words, more refractory patients. The actual responses were higher, 44% versus 19%.
So this was not only statistically significant, but it was a nice numerical difference. Then you can see that continued active therapy increased the A CR responses. These are the a CR twenties to six, nine, and 12 months. But then you show what happens after. You cross over the sham treatment, active stimulation, and they do just as well.
In other words, this therapy clearly effective 'cause it makes a big difference in the sham or placebo treated patients when they cross over as well.
Tiffany: Wow.
Dr. Vibeke Strand: Yeah. And then so you can [00:23:00] see a CR. 50% responses were up to 38% at 12 months. And the a CR twenties were up to 60% at 12 months. And that's really very nice.
And that's as good at 12 months as we've seen with our other therapies.
Tiffany: I also see some ACR seventies there.
Dr. Vibeke Strand: Yep. There are some a CR seventies as well.
Tiffany: Wow.
Dr. Vibeke Strand: So there's, there's a fair amount of improvement and these were. Patients who'd failed at least one, and most of them had failed two TNFs. So this is the safety, and basically they're relatively minor adverse events.
Most of them were procedure related, or to some extent, those who just started stimulation found that it was a little strange when they got the training, but they didn't have any adverse events after that. The serious adverse events [00:24:00] that were related were related to actually the implant procedure, and they were either like hoarseness or problems with the vocal cords.
And it only happened in three out of 122 patients and one out of 120 patients. So didn't happen in very many patients, and the unrelated serious adverse events were due to other causes. So essentially there were no adverse events that led to study discontinuation. Of course, none that led to death, and the majority of them were very mild.
As I said, the ones that were really problematic were because of vocal cord dysfunction, or patients therefore had hoarseness or their VO voice changed dysphonia, and these all improved over time.
Tiffany: Oh, okay. So even those improved.
Dr. Vibeke Strand: They all resolved.
Tiffany: Wow.
Dr. Vibeke Strand: And then this is the [00:25:00] 12 month data. So you can see over 12 months, there were no related serious adverse events.
There were no serious infections, there were no malignancies, there were no cardiac events. And these are. The very patients that we are concerned about when we prescribe our biologics or our targeted synthetics. So basically, I think you would agree with me that this is, this looks like a very safe procedure.
It is a procedure, but it's a one-time event, as it were.
Tiffany: Absolutely. Wow.
Dr. Vibeke Strand: So essentially the setpoint therapy is approved by the FDA and this is the indication. Indicated for use in the treatment of adult patients with moderate to severely active rheumatoid arthritis who've had an inadequate response, loss of response, or intolerance to one or more biologic or targeted synthetic DMARDs.[00:26:00]
And so that means patients who might want a non-drug alternative therapy. Who have had trouble either with compliance or, or continuing to take these therapies who've had side effects or are afraid of the side effects that might occur, and also are afraid of those because of their comorbid conditions, and they've been warned that they should be concerned or they haven't responded to some of these advanced therapies.
So what I wanna tell you is that the vagus nerve is part of the central nervous system. It plays a critical role in balancing inflammation and vagal nerve stimulation works by targeting that nerve and then reduces inflammation similar to our other therapies. But it's a potential new mechanism of action, which is, I would say more [00:27:00] specific.
And the implanted device is effective and safe. I've showed you the data. Patients who've used it have stopped their biologics or other treatments. Some have chosen to use these back in combination with the vagal nerve stimulation, and as we've said, the setpoint system is now approved by the FDA and is indicated.
As I read to you for adults with RA who've had inadequate responses, loss of response, or intolerance to biologic or JAK inhibitors.
Tiffany: So let me, this is absolutely fascinating and one of the reasons is because as a patient myself and in these online communities, this has been a hot topic. I've seen a lot of people sharing about it and.
Mostly all I've seen is it's a, it'll help with your pain. That's the way a lot of patients are talking about it. And [00:28:00] what I learned from you is this is actually a way to treat disease in a different way than. Using the biologics that we're used to. It is a potential option for people. You mentioned some of the people who could be good candidates if they have heart conditions.
Dual diagnosis. I know elderly as well. There's been a lot of, I know as a there, there's a lot of question, how do we treat people with late onset where they ha they're already older and you don't necessarily wanna put 'em on these biologics. There's a lot of people who are afraid of the side effects, to your point.
So I, I have not seen patients talking about these things. So that's a huge takeaway and super, super exciting. I might add, it's
Dr. Vibeke Strand: more than just pain. It definitely does change the disease activity. It definitely also [00:29:00] happens to help the patient reported outcomes. That's gonna be presented soon, I hope. But at any rate.
The data are very, very good. That physical function gets better, pain gets better, global assessment gets better, patients feel better. Their health related quality of life is improved.
Tiffany: Wow. So question then. We, we said it was, it's, it's for approved by the FDA. In the United States, do you know? Is it approved anywhere else or just in the United States?
Dr. Vibeke Strand: Just the United States so far.
Tiffany: Okay. And just for rheumatoid arthritis?
Dr. Vibeke Strand: Just for rheumatoid arthritis. So I know they're gonna be studying some other diseases.
Tiffany: Okay. And if a patient, so this is a avail available now, correct.
Dr. Vibeke Strand: Correct.
Tiffany: Okay. So if somebody listening, a patient listening, wants to talk to their [00:30:00] rheumatologist about this possibility, how do you suggest they approach it?
Dr. Vibeke Strand: Talk to your rheumatologist. Ask your rheumatologist. Okay. And they can always contact the company setpoint and the company can connect them with a neurosurgeon. If they want to do the implantation. So all of that can be arranged. Basically it's, it's approved and it will be reimbursed.
Tiffany: Okay? So we shouldn't have to worry about, oh, well, I'd like to try and now my insurance company is gonna say no.
Is that,
Dr. Vibeke Strand: well, I can't promise you what insurance companies will do. We do know it's been covered by payers.
Tiffany: Okay, well that's, that's good. So, definitely if you're out there and you're trying this and you're talking to your rheumatologist, really would love for you to connect with a arthritis, let us know what are, what has happened.
Where did you get. Access to it. Also, [00:31:00] because this is one of those episodes, because it has the Rheumy rounds part of it where we don't just have patient listeners, we also have health providers who listen to this. So is there anything Vibeke you would say to your peers, to other rheumatologists who may not be familiar with this?
And if a patient comes to them, what would you most want them to know?
Dr. Vibeke Strand: Most of all, I want them to know that they can contact the company setpoint. They will find what they need in terms of how to go about prescribing this medication and using it. And I think it's a very exciting type of therapy. And I think what's also very nice about it is it can be used as a singular therapy or it can be used in combination, but think of the convenience that's associated with it as well.
Tiffany: Absolutely. And as as you said, this is, this can be long term, and I think it's important too for patients as well. Many of [00:32:00] us do have fear of flaring. That's a real thing, right? And I know doctors as well, and so just keep in mind there, this could be. A treat to target type of thing too, where you, you go on it, you're, you're getting better.
You still don't wanna necessarily get rid of everything. But then you realize when you're going in your meeting with your rheumatologist and you see improvements, then you might feel better too about tailoring off or tapering off a treatment. So I think there's just a lot of exciting things. The possibilities here.
So it's super exciting times for an opportunity, an option for patients.
Dr. Vibeke Strand: I think so. I really do. I think this is novel and it is unique.
Tiffany: Absolutely. Well. I found this absolutely fascinating. We definitely want anybody who has participated in the show will provide you with a follow a link, ask more questions, give [00:33:00] us updates.
Have you used this? Have you heard of this? What did you learn the most? What would you still like to learn? We're here to communicate to help make sure that you're informed about new treatments and also how to access them. And we, we want to continue with our patient reported data on how these things used past these clinical trials that were presented today.
Other than that, is there anything else that, that you wanted to add, Vibeke, or did that I
Dr. Vibeke Strand: don't think so. I don't think so, except, thank you.
Tiffany: Absolutely. Thank you so much and for, for everybody listening again, we just wanna thank you for, for coming to the table, listening to this important information on behalf of AiArthritis and the AiArthritis Voices 360 talk show in Rheumy round series.
We are very excited to continue this conversation and thank you again Vibeke so much as always for coming and joining the show.
Dr. Vibeke Strand: Thank you.
Tiffany: All right, thanks everyone.[00:34:00]
Intro: AiArthritis Voices 360 is produced by the International. Foundation for autoimmune and autoinflammatory arthritis. Find us on the web at www.AiArthritis.org. Also, be sure to subscribe to this podcast and stay up to date on all the latest AiArthritis news and events.